Tokyo, Japon

Institutions médicales proposant une médecine régénérative en utilisant des cellules souches
créer un plan de fourniture de médecine régénérative pour chaque technologie de médecine régénérative qu'ils mettent en œuvre, et faites-le examiner pour des raisons de sécurité, efficacité, etc..
par un certifié certifié médecine régénérative commission agréée par le Ministère de la Santé, Le travail et la protection sociale reçoivent.
Institutions médicales proposant une médecine régénérative utilisant des cellules souches、
Créer un plan de fourniture de médecine régénérative pour chaque technologie de médecine régénérative à mettre en œuvre.、
Par le comité spécifique certifié de médecine régénérative agréé par le ministère de la Santé, du Travail et de la Protection sociale.
sécurité、Sera examiné pour l’efficacité, etc.。
Soins de vieillissement (thérapie/injection intraveineuse)
Divers traitements à base de cellules souches tels que la prévention anti-âge
Soins au vieillissement (thérapie/injection intraveineuse)
Divers traitements comme la prévention anti-âge
Tous les membres de notre personnel s'engagent à travailler en étroite collaboration avec vous et à vous prodiguer des soins médicaux au mieux de nos capacités.. Nous avons hâte de vous servir, avec la promesse d'un travail de pointe et d'un accueil de haute qualité.
Proche de tout le monde、Tout notre personnel fera de son mieux pour prodiguer des soins médicaux.。
Nous promettons des compétences avancées et un accueil de haute qualité.、Nous avons hâte de vous voir.。
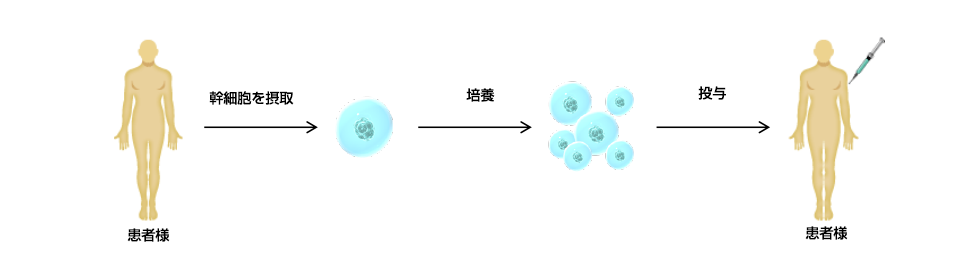
Potentiel des cellules souches mésenchymateuses
Il y a environ 60 des milliards de cellules dans notre corps. Nos mouchoirs, comme le coeur, cerveau, et la peau, sont constitués de cellules qui jouent un rôle extrêmement important dans notre vie quotidienne. Il y a environ 270 types de cellules selon leur rôle. Les cellules qui constituent la base de ces cellules sont « cellules souches. » Les cellules souches sont des cellules qui se transforment en diverses cellules et tissus et existent à des endroits fixes du corps.. Ces cellules produisent des cellules pour remplacer les tissus lorsqu'ils sont endommagés..
On sait depuis longtemps que ces cellules souches existent dans la moelle osseuse.. Cependant, il existe une limite à la quantité de cellules souches pouvant être prélevées dans la moelle osseuse, et la récolte nécessite une anesthésie générale, ce qui est une opération fastidieuse pour les patients. Dans 2000, Docteur. Hédrick de UCLA (Université de Californie, Los Angeles) découvert que la graisse sous-cutanée abdominale contient une grande quantité de cellules souches. Diverses études ont été menées sur les caractéristiques fondamentales des cellules souches mésenchymateuses adipeuses.. Cellules souches mésenchymateuses d'origine adipeuse, qui sont faciles à collecter, très sûr, et ont une large gamme d'efficacité, attirent l’attention en tant que cellules essentielles à la médecine régénérative.
En outre, le « Loi visant à garantir la sécurité de la médecine régénérative, etc.. (Acte n°. 85 de 2013) » a été promulguée en novembre 25, 2014, et recherche clinique (recherche clinique utilisant des cellules souches humaines, etc.) est gratuit. Des règles à respecter par les établissements médicaux et les fabricants de produits de transformation cellulaire ont été établies afin que la recherche, développement, et l'application pratique d'une médecine régénérative de haute qualité peut se dérouler en toute sécurité dans la pratique médicale, et afin de fournir une médecine régénérative de manière rapide et sûre. L'environnement est désormais en place.
Possibilités de cellules souches mésenchymateuses
Il y a environ 60 000 milliards de cellules dans notre corps。cœur、cerveau、Nos tissus, comme notre peau, sont constitués de cellules.、joue un rôle très important dans la vie quotidienne。 Il existe environ 270 types de cellules selon leur rôle.。Les cellules qui constituent la base de ces cellules sont des « cellules souches ».。Les cellules souches sont des cellules qui se transforment en diverses cellules et tissus.、Situé à des endroits fixes dans le corps。Ce sont des cellules qui produisent des cellules pour remplacer les tissus lorsqu’ils sont endommagés.。
On sait depuis longtemps que ces cellules souches existent dans la moelle osseuse.。Cependant, le nombre de cellules souches pouvant être collectées à partir de la moelle osseuse est limité.、Le prélèvement de l’échantillon a nécessité une anesthésie générale, ce qui a nécessité une intervention chirurgicale qui a imposé un fardeau au patient.。2000UCLA en 2016(Université de Californie, Los Angeles)Le Dr Hedrick a découvert que la graisse sous-cutanée abdominale contient de grandes quantités de cellules souches.。Concernant les caractéristiques fondamentales des cellules souches mésenchymateuses adipeuses,、Diverses études ont été réalisées。Facile à collecter、Très sûr、Les cellules souches mésenchymateuses adipeuses ont une large gamme d’efficacité.、Les cellules attirent l’attention comme étant la clé de la médecine régénérative。
Aussi、La « Loi visant à garantir la sécurité de la médecine régénérative, etc. (Loi n° 85 de 2013) » est entrée en vigueur le 25 novembre 2014.、recherche clinique(Recherche clinique utilisant des cellules souches humaines, etc.)、Des règles à respecter par les institutions médicales et les fabricants de produits de transformation cellulaire ont été établies pour garantir que la recherche, le développement et l'application pratique d'une médecine régénérative de haute qualité puissent se dérouler en toute sécurité et avec des soins médicaux gratuits.、L’environnement nécessaire à la fourniture rapide et sûre de médecine régénérative, etc. est désormais en place.。
 Demande d'informations via WhatsApp :
Demande d'informations via WhatsApp :
e-mail: [email protected]
Clause de non-responsabilité: La thérapie par cellules souches est une excellente thérapie alternative mais n'offre aucune garantie et n'est pas présentée comme un remède.. Ceci est similaire à de nombreux autres traitements médicaux conventionnels. Un examen complet des antécédents médicaux du patient est nécessaire pour déterminer l’éligibilité et l’approbation du traitement par cellules souches.. Toutes les informations personnelles fournies sont destinées à un usage interne et médical uniquement auprès des prestataires médicaux.. Les traitements ne sont pas effectués dans des cabinets ou au Royaume-Uni. Toutes les thérapies cellulaires sont dispensées en Europe ou au Japon, car il est réglementé par le ministère de la Santé et d'autres autorités locales. Tous les traitements sont effectués dans les limites légales et le cadre réglementaire du pays dans lequel exerce le prestataire médical concerné..
Les informations présentes sur cette page sont destinées à un usage scientifique, pédagogique, et à des fins d'information générale. Approches cliniques, disponibilité, et le statut réglementaire peut varier selon les pays, institution, et indication médicale. Pour les décisions médicales individuelles, les lecteurs doivent consulter des professionnels de la santé qualifiés et des centres médicaux accrédités.
Cet article a été préparé par l'équipe éditoriale de NBScience dans le cadre de la recherche clinique, biotechnologie, et informations médicales internationales.








